Decades Of Medical Device Design
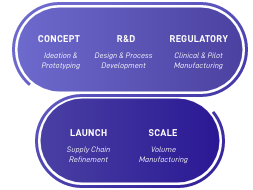
Concept
With a focus on speed and effectiveness, DSA’s material, prototyping, & accelerator services deliver working product quickly.
Design for Manufacturing
Design parts, components, and products with ease of manufacturing at the forefront. We'll discuss your manufacturing strategies early, making recommendations that are scalable and cost efficient.
Quality Systems
Dedicated quality resources will align inspections through test method validations and design verification to scale into commercial manufacturing. Partners use establish quality systems such as ISO 13485 and are FDA registered.
Risk & Cost Optimization
Supply chain assessments are included for all existing and first-time clients, and are an on-going part of the DSA approach. Reduce risk and improve cost efficiency with second sourcing, process improvements, and consolidation.
Supply Planning
Plan distribution, manufacturing, and procurement operations according to demand forecasts. Consider capacity constraints and material availability, manage inventory with safety stock programs, and understand fluctuating lead times for raw material demands.
R&D
DSA will help you align on total cost of device, identifying optimal areas to outsource to design and manufacturing partners.
Design & Manufacturing
Identify the most valuable areas to of your product to outsource to design and manufacturing partners. We'll help you align on total cost of device, performance requirements and consolidation, and then identify vetted partners to deliver a reliable build.
Regulatory Support
DSA supports the development of Quality Management Systems and can help prepare the necessary documentation for regulatory submissions. Our sutures, surgical places, and guidewaires are included in our list of full approved devices.
Second Sourcing
Avoid supply chain interruptions, hold negotiation leverage, and protect your delivery schedules with dual sourcing. Dual procurement is made easier through DSA: we source partners with proven track records and manage purchase orders on your behalf.
COGS Reduction
Product revisions on current and next generation designs give us the opportunity to manufacture less expensive parts. We'll suggest ways to streamline the number of steps, manufacturers, and materials that go in your final product.
Regulatory
DSA reduces risk and improves cost efficiency with second sourcing, process improvements, and consolidation.
Accelerator Services
Medical device accelerator services for early and later stages of design. Universal Handles for benchtop testing and preclinical studies, theoretical modeling software for complex multi-layer shafts, balloon catheters in as little as two weeks, and more.
Testing Services
Component validations, accelerated aging studies, and life cycle fatigue testing of critical implant materials. Meet the experts who can design the right experiments for the necessary data for successful regulatory submissions.
Supply Chain Consolidation
Improve price/part and minimize costs from shipping, inspection, and yield losses by consolidating assemblies. Holistically view the supply chain to identify effective consolidation strategies based from performance, pricing, and location.
Offshore Manufacturing
Our offshore manufacturing facilities are compliant with quality systems, saving cost without sacrificing quality or risking your regulatory approvals. Experienced, compliant with GMP, with clean, consistent record keeping of successful production runs.
Launch & Scale
Get connected with experts & advancements in technical materials, components, & manufacturing processes for Med & Biotech. Our broad network of technical experts is available to attack your challenges and become an extension of your team.
Materials & Services Consulting
Were the materials chosen for proof of concept the best materials for fit and function, cost, and availability? As we move into later R&D stages, we will hone in on your materials selection, ensuring performance, cost, and scalability are at their peak.
Quality Control Systems
Our supply network passes ISO & FDA approvals where needed, ensuring we help you build a supply chain and medical device that will withstand the necessary scrutiny of clinical trials. Access robust manufacturing processes & dedicated quality teams.
Single Point of Contact
Your DSA rep becomes an extension of your team, supporting consolidation, second sourcing, and COGS reduction opportunities. Stay up-to-date with changes in shipping, manage procurement, and streamline your delivery processes.
Supply Base for Acquisition
Each step in the medical device innovation process is for the continuous improvement of a better performing, more cost-effective product. With an eye on exit strategy, we’ll help you your supply base be acquisition ready.
- Structural Heart
- Cardiovascular
- Electrophysiology
- Peripheral Vascular
- Neurovascular
- Mi Orthopedic
- Pulmonary
- Oncology
- Endoscopy
- Laparoscopy
- Urology
- Gynecology


Defining device requirements for R&D Engineers & Project Managers
EXPLORE
1000’s of devices launched, DSA is a go-to resource for competitive advantage.
DEFINE
Defining material and manufacturing requirements form a quick sketch.
TEST
Rapid prototyping prototypes with a specialized network of manufacturers.
REFINE
Set your design apart with seasoned, innovative, engineering experts.

Outsourcing end-to-end development, optimizes in-house manufacturing efforts.
CONTRACT MANUFACTURING
1000’s of devices launched, DSA is a go-to resource for competitive advantage.
FDA REGULATION
Defining material and manufacturing requirements form a quick sketch.
DROPSHIP COMPONENTS
Rapid prototyping prototypes with a specialized network of manufacturers.
EXPAND
Set your design apart with seasoned, innovative, engineering experts.

Source materials with DSA and they’ll arrive fully customized
MATERIALS
- HEAT SHRINK TUBING
- POLYAMIDE TUBING
- STAINLESS STEEL
- NITINOL
- HEAT SHRINK TUBING
- POLYAMIDE TUBING
- STAINLESS STEEL
- NITINOL
- STAINLESS STEEL
CUSTOMIZATIONS
- LABELING & PRINTING
- CUSTOM SIZING
- CUT-TO-LENGTH SERVICES
- LABELING & PRINTING
- CUSTOM SIZING
- CUT-TO-LENGTH SERVICES
- LABELING & PRINTING
- CUSTOM SIZING
- CUSTOM SIZING
For teams who already have the materials they need
BALLOON DESIGN
- BALLOON DEVICE INTEGRATION
- LASER PROCESSING
- LABELING & PRINTING
- CUSTOM SIZING
- CUT-TO-LENGTH SERVICES
- LASER PROCESSING
- LABELING & PRINTING
- CUSTOM SIZING
- CUT-TO-LENGTH SERVICES
LAMINATION ONTO SUBSTRATES
- DELIVERY SYSTEM INTEGRATION
- FABRICATION INTO SYSTEMS
- HEAT STABILIZING
- GRINDING
- FORMING
- FABRICATION INTO SYSTEMS
- HEAT STABILIZING
- GRINDING
- FORMING
Ensuring correct testing, data, and document submission
QUALITY CONTROL
Inspections for every part of the development and commercialization process.
REGULATORY SUPPORT
Helping teams prepare the necessary documents for regulatory submission.
TEST
Designing the experiments needed for regulatory submissions.
ISO & FDA EXPERTS
A supply network dedicated to medical experts. ISO & FDA approvals where needed.
Manufacturing design for risk, cost, and efficacy
RISK & COST OPTIMIZATION
Ongoing supply chain assessments to ensure risk and cost efficiency.
SECOND SOURCING
Avoid supply chain interruptions and hold negotiation leverage.
SUPPLY CHAIN CONSOLIDATION
Improve price/part, minimize costs from shipping & inspection, and yield losses.
SINGLE POINT OF CONTACT
DSA manages consolidation, second sourcing, and COGS reduction opportunities.

Call us!
Call our representatives at the number below, or put in a request form and we will be in contact within 1 business day.
650. 941.0898
Keep up-to-date with the latest in medical device manufacturing
Sign up to receive updates in the field of med tech manufacturing, and receive exclusive consultation offers from seasoned professionals.

Thank you!
A DSA Technical Expert will be in touch to answer your questions in the next 24-48 hours.
Until then, please view our Medical Materials portal. We have compiled thousands of immediately available, in-stock options. All available to inspire your device design, quickly.